Enhanced labeling design and placement
Quva’s ready-to-administer Oxytocin IV bags are updated with redesigned front and back labels, as well as front label placement that covers the diluent bag manufacturers linear barcode so that there is more clarity that Quva’s label barcodes are the ones scanned for the necessary compounded sterile product information like lot number, expiry, etc. This incorporates ISMP’s best practices on responsible oxytocin use, barcode verification, and medication safety.1
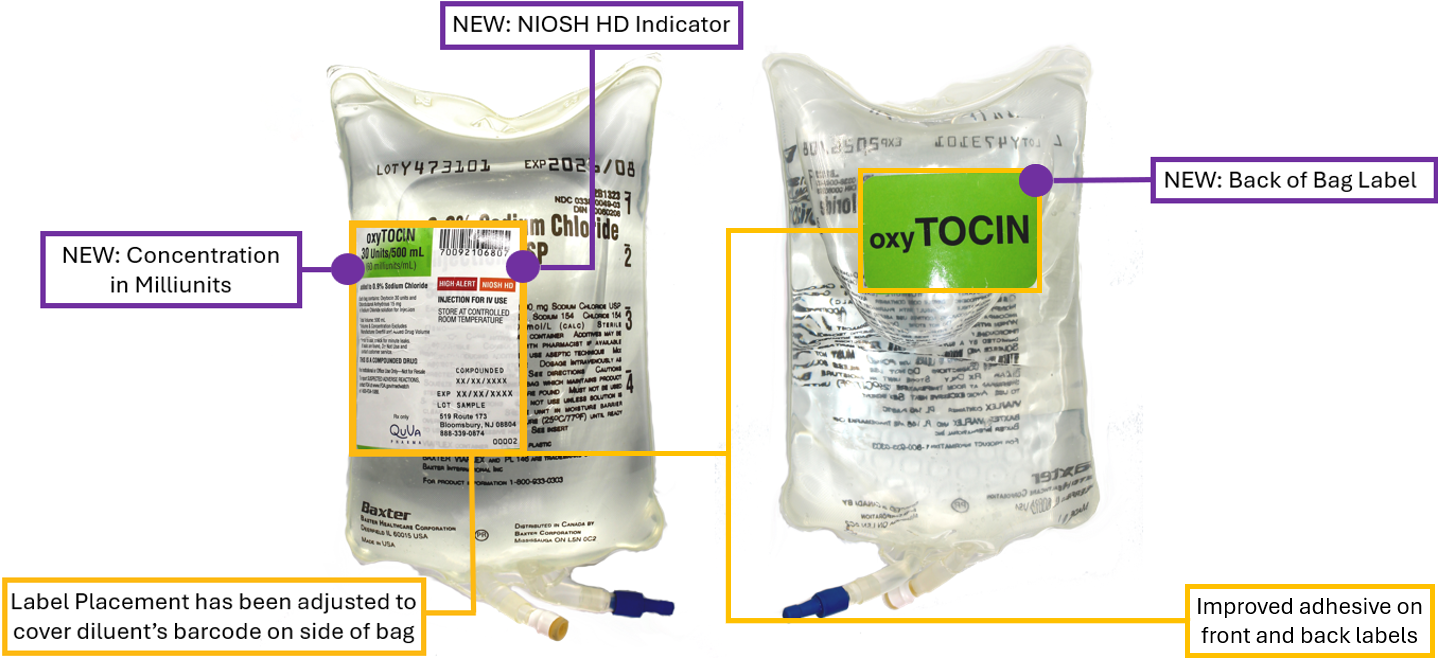

NEW: Front label redesign and enhancements
- Updated label placement and improved adhesive ensures secure coverage of the diluent bag’s barcode, reducing confusion and simplifying workflow by presenting a single barcode and expiration date of the compounded sterile product for scanning
- Concentration expressed in milliunits, supporting more precise titration in clinical settings and aligning with infusion pump dosing nomenclature
- NIOSH HD indicator added to the solution bag, expanding on the current High Alert designation and improving healthcare worker safety
NEW: Back auxiliary label
- Medication name displayed in TALLman lettering on the back of the solution bag, increasing patient safety by aiding clear differentiation of oxytocin IV bags from plain hydrating solutions and magnesium infusions - An ISMP best practice
IV Bag Product specifications
Our oxytocin IV bags are designed to integrate seamlessly into hospital pharmacy operations, supporting safe administration, efficient storage, and consistent availability.
Standardized product details
Each oxytocin IV bag is supplied as a ready-to-administer solution, reducing compounding burden while supporting consistency across care settings.
Key specifications include:
- Oxytocin concentration options commonly used in inpatient settings
- IV bag volumes aligned with standard infusion protocols
- Clearly defined item numbers and packaging configurations
- Lot-level traceability to support inventory management and recalls
Detailed product-specific information, including item numbers and packaging details, is available upon request or within accompanying product documentation.
Storage and handling requirements
Oxytocin IV bags are manufactured, packaged, and labeled to support safe handling throughout the medication lifecycle from receipt through administration.
Storage conditions
- Stored under controlled conditions consistent with labeled requirements
- Designed to maintain stability throughout the stated shelf life when stored as directed
- Clearly labeled to support pharmacy inventory control and compliance
Handling considerations
- Ready-to-administer format reduces preparation steps in the pharmacy
- Barcoded labeling supports bedside scanning and closed-loop medication workflows
- Packaging aligns with hospital policies for safe transport and storage
Focusing on Compliance, Safety, and Convenience
Purchasing ready-to-administer oxytocin is a key strategy employed by hospitals and health systems, with 56% of hospital pharmacies outsourcing oxytocin due to difficulties in compounding.2
Our products help:
- Reduce the risk of exposure to healthcare personnel, patients, and environment
- Satisfy both USP 797 compounding regulations and USP 800 regulations for handling hazardous drugs3
- Saves time and cost associated with compounding
Quva Pharma's Oxytocin Products
(DAYS)
QUANTITY
Put patient safety first with our ready-to-administer Oxytocin products today!
DOWNLOAD PRODUCT INFORMATION SHEET
References
- 2024-2025 ISMP Targeted Medication Safety Best Practices for Hospitals. https://online.ecri.org/hubfs/ISMP/Resources/ISMP_TargetedMedicationSafetyBestPractices_Hospitals.pdf
- Pharmacy Purchasing and Products. Outsourced Compounding. https://www.pppmag.com/article/2911
- USP. Hazardous Drugs—Handling in Healthcare Settings. https://www.usp.org/compounding/general-chapter-hazardous-drugs-handling-healthcare
